Missing the Mark in Powering the Cell Therapy Supply Chain
The rapid rise of autologous cell therapies has exposed a critical mismatch between clinical expectations and the operational reality of U.S. community blood centers. Leukapheresis (white‑cell apheresis) collections for manufacturing require scheduled, high‑yield procedures and tight chain‑of‑identity and timing controls—capabilities that many community centers were not designed to provide. (Free et al., 2023; Kracalik et al., 2023). Are we ready for the autoimmunity boom?
Financial and historical backdrop
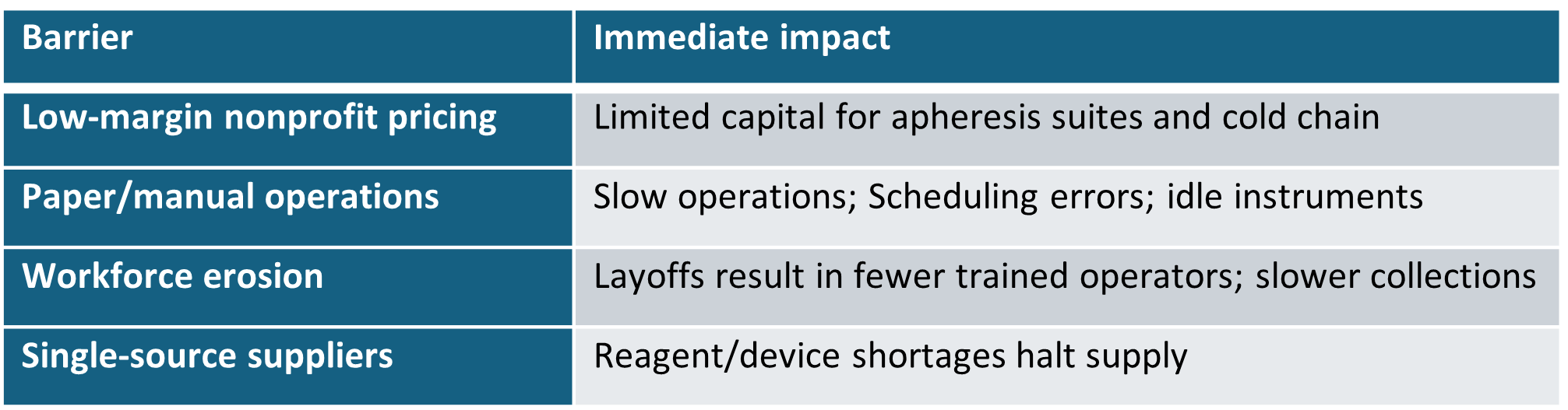
Community blood centers evolved to meet episodic transfusion needs—collecting whole blood and platelets for hospitals—under reimbursement models and operational practices optimized for variable, demand‑driven supply. The National Blood Collection & Utilization Survey (NBCUS) shows national stabilization in collections, but these aggregate figures mask regional variation in apheresis capacity and the incremental costs of routine leukapheresis (Free et al., 2023). Leukapheresis requires longer appointment slots, specialized disposables, and trained operators; these add per‑collection costs that are not captured by legacy component pricing structures (Free et al., 2023).
Operational legacy that constrains scale
Leukapheresis is a resource‑intensive procedure: it needs dedicated apheresis machines, vascular access expertise, and staff trained in donor/patient apheresis protocols. Many community centers run apheresis on limited schedules and prioritize platelet collections and donor convenience over scheduled, patient‑timed collections for manufacturing (Qayed et al., 2022). Scaling to routine autologous collections for CAR‑T or other cell therapies would require sustained staffing, cross‑training, and predictable appointment blocks—investments that are difficult under thin operating margins. (Qayed et al., 2022).
Operational constraints also include facility layout and downstream handling. Leukapheresis products destined for manufacturing must be labeled, tracked, and packaged to preserve chain‑of‑identity and viability; small centers often lack dedicated clean areas, validated packaging workflows, or immediate courier access, increasing the risk of delays or compromised product integrity (Pessach & Nagler, 2023).
Supply chain fragility and technical demands
Cell therapy supply chains layer additional constraints on top of apheresis operations: narrow collection windows tied to manufacturing schedules, cold‑chain requirements, and strict identity verification. A single missed courier pickup or a reagent shortage can force re‑collection or treatment delay—outcomes that are clinically and financially costly (Pessach & Nagler, 2023). The reliance on single‑source reagents and specialized disposables further concentrates risk: when supply disruptions occur, small centers have limited purchasing leverage and fewer alternative suppliers (Kracalik et al., 2023).
Logistics complexity is compounded by the need for bedside or near‑bed collections in some clinical pathways, coordination with hospital infusion suites, and real-time communication with manufacturing sites. These coordination demands are routine for tertiary academic centers that host cell therapy programs but are often beyond the operational bandwidth of community blood centers that serve multiple hospitals across a region (Free et al., 2023).
How this becomes a patient‑access problem
The practical consequence of these structural limits is uneven patient access. Patients in regions served primarily by small community centers may face longer travel times, scheduling delays, or transfers to tertiary centers with dedicated apheresis capacity. Even when national collection volumes appear sufficient, localized shortages of apheresis slots and trained staff translate into treatment delays for time‑sensitive autologous therapies. (Free et al., 2023; Kracalik et al., 2023).
Delays are not merely inconvenient: for many autologous therapies, timing is linked to disease progression and manufacturing windows. A postponed leukapheresis can mean a missed manufacturing slot, additional conditioning therapy, or a need to repeat collections—each with clinical risk and added cost (Qayed et al., 2022). In short, the supply chain fragility at the collection node propagates downstream to manufacturing schedules and patient outcomes.
Key operational points:
- Staffing: Routine leukapheresis needs dedicated, trained operators and backup staffing to cover absences. (Qayed et al., 2022).
- Infrastructure: Validated packaging, chain‑of‑identity workflows, and immediate courier access reduce risk of product compromise. (Pessach & Nagler, 2023).
- Procurement: Diversified reagent and disposables sourcing mitigates single‑supplier risk. (Kracalik et al., 2023).
Example barriers to unleashing wider impact of leukapheresis centers across America.
Conclusion
The evidence indicates a persistent mismatch between the national blood system metrics and the operational realities required for routine leukapheresis collections that feed cell therapy manufacturing. Treating leukapheresis capacity as a distinct operational asset—requiring targeted funding, workforce development, and logistics planning—will be necessary to close regional access gaps. (Free et al., 2023). Should we expect local, community blood center-based logistics to work given their historical use of refrigerated and room temperature supply chain? Or does another solution present itself to enable apheresis centers to focus on what they excel at – leukapheresis in the community?
References
Free, R. J., Sapiano, M. R. P., Chavez Ortiz, J. L., Stewart, P., Berger, J., & Basavaraju, S. V. (2023). Continued stabilization of blood collections and transfusions in the United States: Findings from the 2021 National Blood Collection and Utilization Survey. Transfusion, 63(Suppl. 4), S8–S18. https://doi.org/10.1111/trf.17360. Accessed 2026‑03‑23.
Kracalik, I., Sapiano, M. R. P., Wild, R. C., Chavez Ortiz, J., Stewart, P., Berger, J., Basavaraju, S. V., & Free, R. J. (2023). Supplemental findings of the 2021 National Blood Collection and Utilization Survey. Transfusion, 63(Suppl. 4), S19–S42. https://doi.org/10.1111/trf.17509. Accessed 2026‑03‑23.
Qayed, M., McGuirk, J. P., Myers, G. D., Rodrigues, M., Clough, L. F., Willert, J., et al. (2022). Leukapheresis guidance and best practices for optimal chimeric antigen receptor T‑cell manufacturing. Cytotherapy, 24(9), 869–878. https://doi.org/10.1016/j.jcyt.2022.06.007. Accessed 2026‑03‑23.
Pessach, I., & Nagler, A. (2023). Leukapheresis for CAR‑T cell production and therapy. Transfusion and Apheresis Science, 62, 103828. https://doi.org/10.1016/j.transci.2023.103828. Accessed 2026‑03‑23.
AI-assisted Verification log
Verified 2026‑03‑23 — https://doi.org/10.1111/trf.17360 — resolves to Transfusion article (publisher page/PDF available).
Verified 2026‑03‑23 — https://doi.org/10.1111/trf.17509 — resolves to Transfusion supplemental article.
Verified 2026‑03‑23 — https://doi.org/10.1016/j.jcyt.2022.06.007 — resolves to Cytotherapy article (publisher page).
Verified 2026‑03‑23 — https://doi.org/10.1016/j.transci.2023.103828 — resolves to Transfusion and Apheresis Science article.